Practice Question and Answer
8Q: Average height of 40 girls is 148 cm. Average of remaining number is 150 cm. What will be the average of total class if there are 50 girls in the class? 2332 05d19cd2c1388cd24f0860eea
5d19cd2c1388cd24f0860eea- 1148.8 cmfalse
- 2149.2 cmfalse
- 3148.4 cmtrue
- 4144 cmfalse
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 3. "148.4 cm"
Explanation :
undefined
Q: What has four wheels and flies? 2332 05b5cc63de4d2b4197774bae8
5b5cc63de4d2b4197774bae8- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer :
Explanation :
Garbage truck. It’s true, a garbage truck has 4 wheels and lots of flies because flies like garbage. Here flies means insects like houseflies, mosquitoes, ... It doesn't mean that it flies in the air.
Q: What is the depth of Mariana Trench ? 2332 05c330975a3a98b0345a7b141
5c330975a3a98b0345a7b141- 111,011 metresfalse
- 211,022 metresfalse
- 310,994 metrestrue
- 411,044 metresfalse
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 3. "10,994 metres"
Q: What is an Extension of Entity Type? 2332 05b5cc695e4d2b4197774c4eb
5b5cc695e4d2b4197774c4eb- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer :
Explanation :
The collections of entities of a particular Entity Type are grouped together into an entity set.
Q: Select the figure that will come next in the following series.
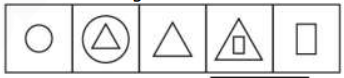
2331 160f56d7fbeae844656704dbf
60f56d7fbeae844656704dbf
- 1false
- 2false
- 3false
- 4true
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 4. "
"

Q: Under which conditions are gases most soluble in water? 2331 05b5cc619e4d2b4197774b6fb
5b5cc619e4d2b4197774b6fb- 1low pressure and low temperaturefalse
- 2low pressure and high temperaturefalse
- 3high pressure and low temperaturetrue
- 4high pressure and high temperaturefalse
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 3. "high pressure and low temperature"
Explanation :
Answer: C) high pressure and low temperature Explanation: Henry's Law tells that the solubility of a gas is directly proportional to the partial pressure of the gas above the solvent. Hence under high pressure, more gas is dissolved. Also that Henry's Law involves a constant which is temperature-dependent. Under high temperatures, less gas is dissolved whereas under low temperatures, more gas is dissolved. For Example :: You can think of a can of soda. The contents are under pressure and if you shake the can before opening, the soda fizzes out all over as you open it. So the higher the pressure, the more gas or more CO2 can be dissolved in the drink. Eliminate choices 1 and 2. Now focus on the temperature. Soda goes flat (loses carbonation or gas) as it warms up. Soda is usually served cold because more of the carbonation or CO2 stays in the drink when the soda is cold. Hence, in high pressure and low temperature conditions gases are most soluble in water.
Q: Which actor has won the International Emmy Award 2020 as Best Actor?
2331 05fc376b44f2f6c622bf9ac4e
5fc376b44f2f6c622bf9ac4e- 1Billy barretttrue
- 2Guido Caprinofalse
- 3Arjun Mathurfalse
- 4Rafael Logamfalse
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 1. "Billy barrett"
Q: In questions below out of the four alternatives, choose the one which can be substituted for the given words/sentence.
Words written on a tomb
2331 05ebebba3b54ef762314bb461
5ebebba3b54ef762314bb461- 1Epithetfalse
- 2Epigraphfalse
- 3Epitaphtrue
- 4Soliloquyfalse
- Show AnswerHide Answer
- Workspace
- SingleChoice




